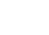Tomasz Płaska
Published on 2020-08-18
Pharmaceutical quality of raw materials is an advantage that manufacturers like to emphasize and should be proud of. Although it is not necessary for the e-liquids industry, some manufacturers want to surpass the quality standards. Therefore, many e-liquid manufacturers use pharmaceutical quality raw materials (like pure nicotine, PG and VG) in their production. In this article, we take a closer look at what you. as a manufacturer or brand owner, should know about the pharmaceutical quality of e-liquids and raw materials. Learn how to check the product’s quality is as high as you expect.
See also:
- OEM Services And Private & White Label: 9 Reasons To Choose Chemnovatic As The Best Business Partner
- Brand Owner: How To Develop Your Own E-Liquid Brand?
- What Are E-Liquids? History And Business Opportunities
- DIY E-Liquids Making: How To Start?
- Natural Vaping Products: Bases, Salts, Flavorings
- Safe Usage Of Chemicals [Guide]
- The Best E-Liquid Flavors To Make Your Recipes Stand Out
- 4 Features Of Nicotine Salts You May Benefit From
- All You Need To Know About E-Liquid Additives
- What Are Nicotine Pouches? 8 Facts You Didn’t Know Yet
Before we start, we have to explain what is pharmacopeia and why is it so important.
Pharmacopeia – What Is It, And What Does It Describe?
With the development of pharmacy in the 16th century, there was a need to create a uniform, official list of substances. That list would guarantee the appropriate quality of chemicals.
In this way, the first pharmacopeia began to appear. According to the definition, it is “an official list of drugs, containing binding standards for their composition, dosage, preparation, storage, as well as methods of testing their quality and evaluation”.
To clarify, pharmacopeia is a reference point for chemical substances in the pharmaceutical industry. In other words, it is the main source of information for chemists and pharmacists regarding the purity, quality, preparation, and application of chemical substances like drugs, vaccines, antibiotics, etc.
Many countries have developed their own pharmacopoeial standards and own pharmacopeias. Nevertheless, the two main and most general reference points for chemists are European pharmacopeia (Ph. Eur. /EP) and American pharmacopeia (USP).
Pharmaceutical Quality Of E-Liquids And Raw Materials: What Does It Mean?
The term “pharmaceutical quality” means that a given substance is of pharmacopeia standards. However, before being approved for production in the pharmaceutical industry, a given substance needs to go through complicated way of pharmaceutical formal regulations. In this respect, not every substance of “pharmacopeia quality” can be used in pharmacy.
To sum up and avoid any misunderstandings: although some raw materials for e-liquid production are of pharmaceutical quality, they usually can’t be named as pharmaceutical grade raw materials.
Raw materials used in the production of e-liquids do not need to comply with pharmacopeia standards. Nevertheless, many e-liquid manufacturers use pharmaceutical quality raw materials like pure nicotine or PG/VG. It is because of the fact, that the most conscious and professional manufacturers want the best quality for their potential customers. It is out of concern for the welfare and health of e-liquid users.
Check also:
- E-Liquid Manufacturing Standards: Tips For Manufacturers And Brand Owners
- PG/VG Ratio: How To Find The Best Ratio For E-Liquid?
- E-Liquid Manufacturing Best Practices: Do You Know Them All?
- What Are Nicotine Toothpicks?
- NicSalts vs. NicShots: Nicotine Salt Shot And Freebase Nicotine Shot Comparison
- Nicotine Pouches: Key Statistics
- Does Higher Nicotine Content Lead To Better Nicotine Absorption?
Pharmaceutical Quality Of E-Liquids: How To Check The Product And Materials Quality?
We know how much you want to give your customers only the best products, so be sure to read our checklist and short tutorial on “decrypting” documents and certificates.
1. How To Read Product Certificates (MSDS, CoA, CoQ, TS, DS)?
So much for the theoretical knowledge, but how to check If an ingredient is of pharmaceutical quality, or not? Let’s say, you are an e-liquid manufacturer and would like to buy pharmaceutical-quality pure nicotine. First of all, always ask your supplier for Safety Data Sheet and other product certificates (which names may vary between different companies). At Chemnovatic we provide the following documents:
- MSDS (Safety Data Sheet) for all of our products,
- CoA (Certificate of Analysis) for pure nicotine,
- TS (Technical Specification) for PG and VG,
- CoQ (Certificate of Quality) for e-liquid bases,
- DS (Data Sheet) for flavours.
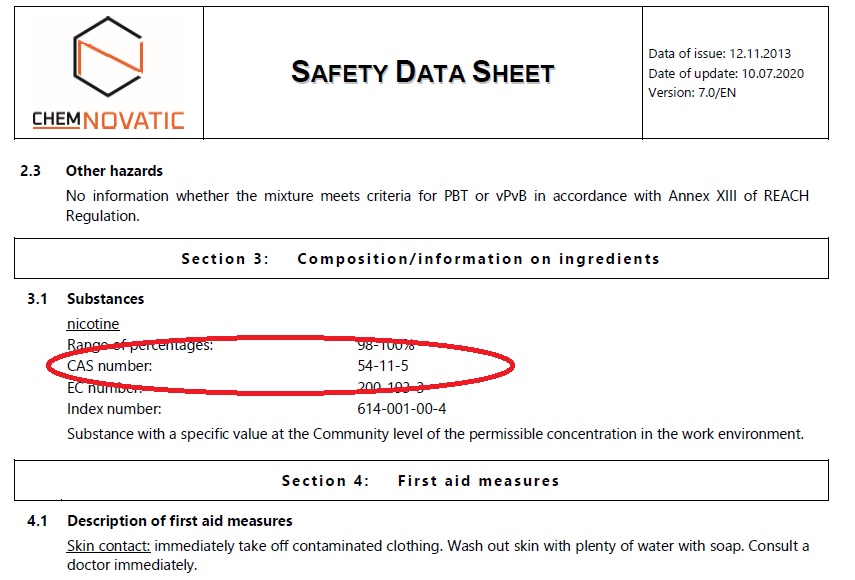
Another important element of ensuring the products’ quality is its composition. Be sure to ALWAYS check the CAS number! It is an identifier of a substance according to the Chemical Abstracts Service (CAS). Moreover, even in a situation when a manufacturer gives a misleading commercial name to its product, CAS number will never lie to you. You can easily Google it and check whether the product is REALLY 100% what you expect it to be.
Moreover, you can also check CAS number authenticity at the European Chemicals Agency (ECHA). In addition to the CAS itself, you can obtain information on the registration of these substances, their CLP classification, and others. Below, you can see an example of how CAS number can be presented:
You can see CAS number of nicotine which is 54-11-5. Moreover, from MSDS you can learn possible threats posed by a given substance, first-aid measures, and precautions to be taken. It is important for ensuring the safety of employees. MSDS also contains information on the storage and handling of a given product. If you would like to know more about storing and handling, we have published a vast article with many interesting details. Be sure to read it here.
2. How To Read Analytical Reports?
MSDS and other documents (CoA, CoQ, TS, DS) are very important. However, these are internal documents that each enterprise prepares individually.
How about checking the quality of products by a third party? It is a good idea to prove the quality of the products by presenting laboratory tests. A reliable supplier is able to provide laboratory analysis of a given product made in an external, independent laboratory.
At Chemnovatic we test our flagship product PureNic 99+ pure nicotine in external, accredited laboratories. We test parameters of pure nicotine in terms of the European pharmacopeia (EP) and American pharmacopeia (USP) standards.
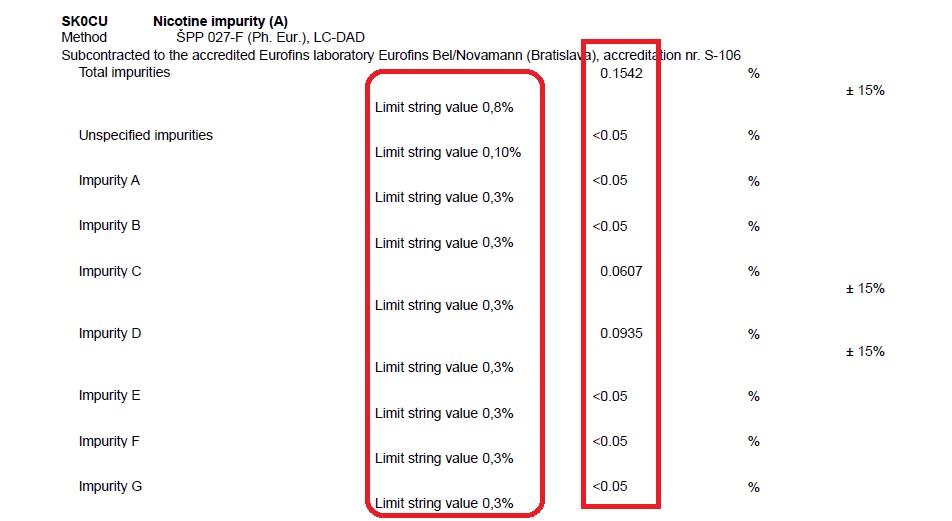
Analytical report for PureNic 99+
We test such parameters as water content, specific optical rotation, appearance, nicotine content, impurities, and heavy metals content. Testing all these parameters, and methods of testing are specified in Eur. Ph. and USP. However, describing all the methods of testing require specific chemical, complicated jargon.
Nevertheless, we can and we SHOULD explain the results of the report. Let’s take a closer look at the first parameter tested, which is water content.

You can clearly see that Chemnovatic PureNic 99+ result is 0,08% as highlighted. Whereas the acceptable content of water in pure nicotine is 0,5% for USP, and from 0,5% for EP. You can see that content of water in Chemnovatic pure nicotine is even lower than the acceptable level specified in pharmaceutical standards.
In other words, in terms of tested parameters, Chemnovatic pure nicotine complies with pharmacopeia standards. Although our nicotine is not intended for use in pharmaceutical industries, it meets the qualitative requirements of the pharmacopeia in terms of the tested parameters confirming its identity and purity level.
Learn more:
- What Is Nicotine And How Is It Classified?
- From Leaves To Labs: A Brief Story About Nicotine Production
- Hard To Handle, Easy To Vape: How To Work With Nicotine Safely?
- Storing And Handling Pure Nicotine & Nicotine Salts
3. Compare the analytical reports with products’ documentation
Another parameter that requires explanation is the content of impurities. As you can see, for each impurity there is a specified certain limit (Limit string value). Next to it, you can see the result of testes PureNic 99+ sample. As you can see, each impurity is way below the pharmacopeia limit.
Importantly, the contaminants tested in the analytical report marked with letters A to G are identified in the CoA document. It is pictured as follows:
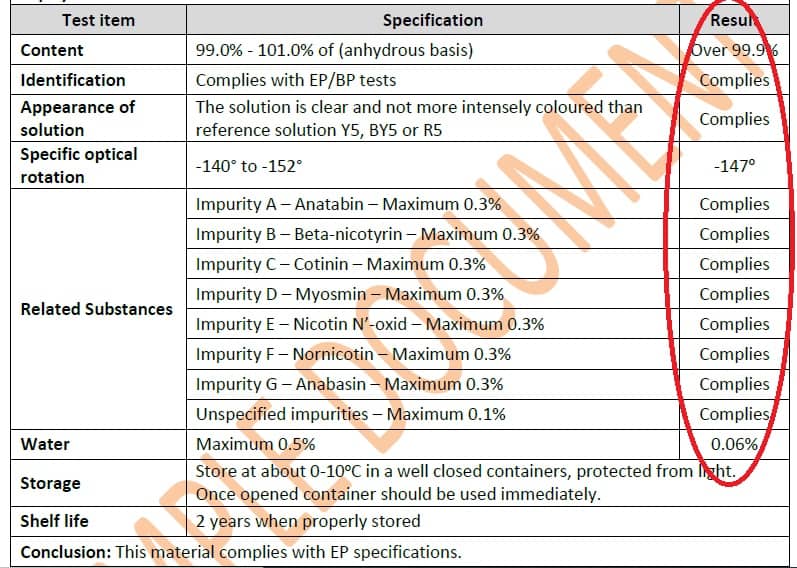
On the basis of the two above examples, you can go step by step and read the full analytical report on PureNic 99+ pure nicotine parameter by parameter. We encourage everyone to download and to analyze it!
Moreover, we can guarantee PureNic 99+ if of pharmaceutical quality with respect to every parameter.
However, if you have any questions, or would like to discuss the report with our advisors, you can contact us directly at sales@chemnovatic.com.
Top-Quality Nicotine Bases: What You Should Know?
When buying semi-finished products for e-liquids, it is worth paying attention to the quality of the raw materials. Propylene glycol (PG) and vegetable glycerine (VG) that suppliers provide should also be in compliance with USP/EP standards. You can check its quality on the basis of MSDS and CoA. Unfortunately, not every producer of raw materials can boast of the pharmaceutical quality of their products.
Probably the worst thing any e-liquid manufacturer can do is to waste great quality pure nicotine and mix it with subpar, low quality, cheap VG or PG.
At Chemnovatic we use ONLY the purest vegetable glycerin and propylene glycol available on the market to act as a nicotine carrier liquid in our VG, PG, and nicotine bases.
Please feel free to check our certificates for VG, PG, and pure nicotine here.
See also:
- What Is Nicotine Base And How To Use It?
- How To Choose The Perfect E-Liquid Base?
- The Differences Between Glycol And Glycerine: When To Use Which?
- 5 Steps To High Quality Nicotine Bases
- Mix Your First E-Liquid Using DIY Bases
- What Makes A Good Nicotine Base?
Chemnovatic nicotine bases
For Chemnovatic top quality of products has always been a priority. With us, you have a guarantee that the raw materials (PG, VG, and pure nicotine) used for the manufacturing of nicotine bases, are of pharmaceutical quality. Moreover, we confirm it by various certificates. You don’t need to believe what we say, because you can check it on your own – just click the button.
Production of Chemnovatic e-liquid bases takes place in a professional, clean production facility to avoid any cross-contamination and impurities getting into our bases.
Additionally, the manufacturing process of nicotine bases eliminates oxygen contact during mixing, pouring, and capping/ sealing the final product. This reduces nicotine oxidation to an absolute minimum.
As a result, our nicotine bases are of the highest possible quality. Thanks to it, they will serve as a great source of nicotine for the e-liquid you are manufacturing.

Want to start your e-liquid brand? Contact us rapidly!
Do you have any questions regarding the quality of our products? Our advisors will help you with the pleasure to read specific parameters on our internal documents and analytical reports regarding our products.
Please, contact us at sales@chemnovatic.com

Tomasz Płaska ✉
Head of Marketing at Chemnovatic, with over 10 years of experience in regulated industries across B2B and B2C segments. At Chemnovatic, he is responsible for building the B2B brand in international markets, demand generation, and e-commerce sales. Previously, he gained experience in sectors including retail chains, analytics, and finance.
Let’s grow your business together!
Subscribe to our newsletter and receive a free access to our e-mail course
on raw materials for e-liquids production (and more!).
No spam, only valuable content we promise to send you.